Power-to-X technologies and renewables: Generating Hydrogen, Saving on Electricity
ZSW looking into biomass combustion combined with high-temperature electrolysis
Storage solutions will be very much in demand as renewables account for a growing share of electricity in the grid. One option – converting fluctuating green electricity into chemical energy carriers or raw materials – looks particularly promising. Scientists at the Centre for Solar Energy and Hydrogen Research Baden-Württemberg (ZSW) want to put power-to-X processes into action with a two-pronged strategy. They plan to combine high- temperature biomass combustion with high-temperature electrolysis to reduce the amount of electricity it takes to produce renewable hydrogen, the feedstock for all chemical power-to-X storage media. The ZSW researchers expect that this could slash overall electricity consumption by half. The first preliminary tests met with success and the response to the project has been positive.
The Working Group on Energy Balances says the share of renewable energies in Germany’s electrical power yield averaged around 36 percent in 2017. The Federal Network Agency’s SMARD information platform noted that its share soared to 100 percent for a few hours each on January 1 and May 1, 2018. The German government is shooting for an average of 65 percent of green electricity to flow into the grid by 2030. This means that the supply of renewable electrical power will increasingly outstrip demand.
From time to time, alternative sources will produce double – and then some – the amount of electricity needed in the grid. All this excess power will have to be converted, so smart conversion solutions will have to be found.
The feedstock for all power-to-X processes – hydrogen
“Power-to-X” is a synonym for promising long-term storage solutions. This blanket term covers all processes that convert green electricity into chemical energy carriers – that is, into electricity-based fuels for vehicles or raw materials for the chemical industry. Power-to-X processes provide a cleaner, climate-friendlier way of producing hydrogen for fuel-cell vehicles, methane for natural gas cars, kerosene for aircraft, liquefied natural gas (LNG) for ships, and basic chemicals for the chemical industry.
Hydrogen is produced from water by electrolysis using green electricity. It serves as the feedstock for all power-to-X technologies. The ZSW is now setting out to combine high-temperature electrolysis and oxyfuel combustion to produce renewable hydrogen in a way that con- sumes far less electricity.
Heat for electrolysis, oxygen for combustion
Alkaline and PEM electrolysis require a lot of electrical power. This is not the case with high-temperature electrolysis, especially when heat replaces much of the electricity needed as the input energy. The oxy-fuel process that provides this heat burns oxygen far more efficiently than purely air-based combustion processes. The researchers use wood or biomass residue for fuel.
Electrolysis, in turn, produces the oxygen required for high- temperature combustion, which normally consumes a great deal of energy. “We aim to generate one cubic meter of hydrogen from 2.5 kilowatt hours of electricity with this technology,” says Dr. Michael Specht, head of ZSW’s Renewable Energy Sources and Processes department. Today’s electrolyzers generally require around twice as much electrical energy.
But these scientists aim to take this a step further by applying the elec- trolyzed hydrogen to convert the “green” carbon dioxide produced by oxy-fuel combustion into a carbon-based energy carrier such as me- thane or into basic chemicals such as methanol. The method uses carbon very efficiently and does not produce more carbon dioxide. And it saves energy because carbon dioxide does not have to be separated from a flue gas.
Looking into two concepts for reactors
The research team is investigating and comparing two types of reac- tors that are to help achieve these goals, one being a fluidized bed reactor and the other a FLOX burner for flameless oxidation. The idea is to generate an oxygen-poor exhaust gas stream that provides key components – the high-temperature heat needed for electrolysis and the carbon dioxide required for subsequent synthesis. Initial trials that used natural gas-fired oxy-fuel combustion in the FLOX burner provid- ed a hot exhaust gas that is well-suited for subsequent fuel synthesis.
Scientists are also simulating this new power-to-x path to assess it. An initial analysis conducted by the Karlsruhe Institute of Technology’s Institute of Technology Assessment and Systems Analysis (ITAS) confirmed that the ZSW’s concept has the potential to significantly reduce the carbon footprint with a relatively low amount of overall energy ex- penditure.
The ZSW scientists are now building a test bed to investigate the combination of these two technologies. “We also want to win over industrial partners that develop high-temperature electrolysis methods for our project,” says Specht.
Funding from the Federal Ministry of Education and Research
This research project is initially scheduled to run for three years with the Federal Ministry of Education and Research (BMBF) pitching in over €900,000 (funding code 03SFK2C0). It is based on the work that the ZSW is doing for the BMBF’s Copernicus project, a power-to-X initiative that is to run for ten years: www.kopernikus-projekte.de/projekte/power-to-x
About the Zentrum für Sonnenenergie- und Wasserstoff-Forschung
The Zentrum für Sonnenenergie- und Wasserstoff-Forschung Baden-Württemberg (Centre for Solar Energy and Hydrogen Research Baden-Württemberg, ZSW) is one of the leading institutes for applied research in the areas of photovoltaics, renewable fuels, battery technology, fuel cells and energy system analysis. There are currently around 250 scientists, engineers and technicians employed at ZSW’s three locations in Stuttgart, Ulm and Widderstall. In addition, there are 90 research and student assistants.
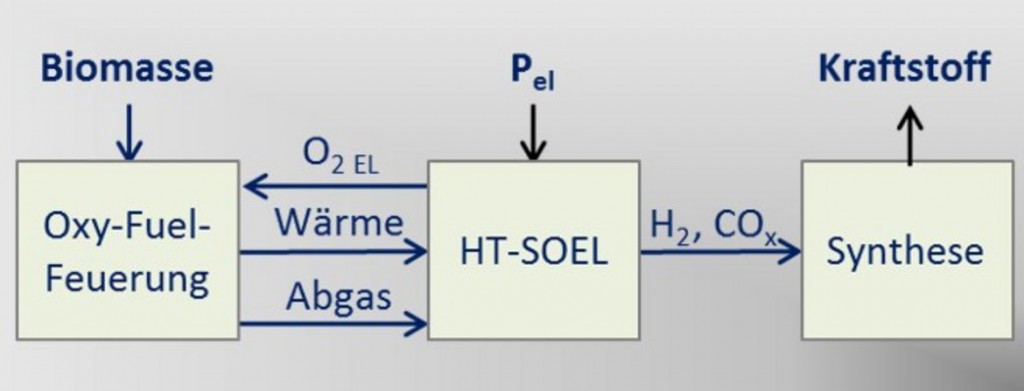
This is how scientists aim to combine an oxy-fuel process with high-temperature electrolysis.
Source: Centre for Solar Energy and Hydrogen Research, press release, 2018-08-01.
